November 17, 2020
Relaxing cell divisions
Scientists at IST Austria discovered how tissue tension controls cell division
Watch the corresponding video on YouTube
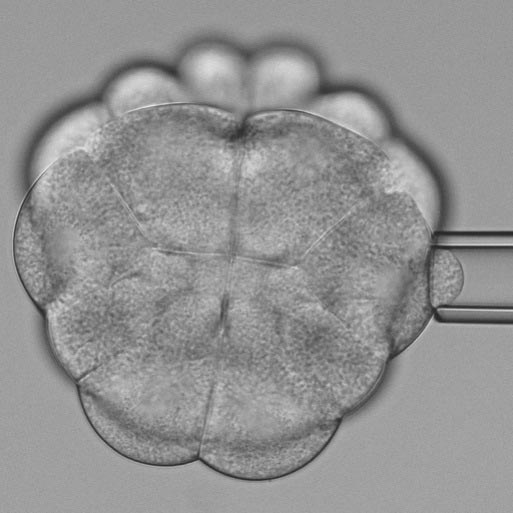
An ascidian embryo at the 32-cell stages. The asterisks mark soft cells at the bottom, which enter cell division and orient their division plane perpendicular to the non-dividing cells at the top by which they are pulled and deformed. © Benoit Godard / IST Austria
During one lifetime, the human body experiences ten quadrillion cell divisions. This biological process is essential to form and maintain tissues and organs within the body. Now, Professor Carl-Philipp Heisenberg and his team at the Institute of Science and Technology Austria discovered how mechanical tension from surrounding tissue influences the division process. The scientists published their study in the journal Developmental Cell. This study presents an entirely new influence on cell division and could also be important for tumor research.
During cell division, a parent cell divides into two daughter cells. Their new role and function depend on the orientation of the division plane. A crucial factor guiding this division orientation is the shape of the mother cell. Now, Professor Carl-Philipp Heisenberg and Benoit Godard, a Postdoc in the Heisenberg lab, discovered – in collaboration with the McDougal lab at Sorbonne University – that the dividing cell softens and thus becomes deformable by mechanical tension originating from neighboring cells, thereby explaining how tissue tension influences cell division orientation.
“The neighboring tissue pulls and deforms the soft dividing cells. Consequently, this alters its cell division orientation and, therefore, the fate of its daughters,” explains Carl-Philipp Heisenberg.
A new model organism
Investigating cellular interactions within a tissue is challenging. Due to the large number of cells involved, cause and effect are hard to disentangle. Therefore, Carl-Philipp Heisenberg utilized a unique model organism. Ascidians, small marine invertebrates, develop nearly identical to vertebrates but only consist of very few cells. Additionally, the cell’s fate is set quite early. With fewer cells and a predetermined development, ascidians allow scientists to investigate the tissue dynamics more precisely.
© Benoit Godard / IST Austria
Observing dynamics cell shaping
It is known for more than a century that cells tend to divide perpendicular to their long axis. Previously, cells undergoing division were thought to be stiff and thus insensitive to mechanical forces from their environment affecting their shape and thus division orientation. Opposite to these beliefs, Carl-Philipp Heisenberg and his team now discovered that dividing cells can become softer and thus deformable by extrinsic forces.
“We had preliminary and indirect results suggesting that dividing cells become soft, but this was opposite to the literature on cell division. Thus, when I conducted the biophysical measurement of cell stiffness and saw the softening occurring, it was the greatest moment in my scientific career,” remembers Benoit Godard.
By performing a three-dimensional scan of the cell shapes in the developing ascidian embryos and combining it with biophysical measurements of cell stiffness, the scientists found that dividing cells became softer and, consequently, deformed in response to forces from neighboring cells.
“We wanted to identify the governing factors of cell division. We found that cells become soft when undergoing division and that this allows extrinsic mechanical forces to influence the shape and thus division orientation of the dividing cell,” summarizes Carl-Philipp Heisenberg.
This study opens the path to novel fundamental research but might also be relevant for clinical studies. For example, tumors change tissue tension, which again likely influences tumor cells’ rate and division orientation. These results help to explain tissue malformation and might enable external controlled tissue formation.
Publication
Godard et al.. 2020. Apical relaxation during mitotic rounding promotes tension-oriented cell division. Developmental Cell. DOI: 10.1016/j.devcel.2020.10.016
Funding information
This project was jointly funded by the FWF and the French state funding agency Agence National de la Recherche (ANR “MorCell”: ANR-17-CE 13-002 8).



