January 11, 2024
Synapses Brought to the Point
Scientists unravel structure and function of important inhibitory synapses in the cerebellum
Whether picking up a small object like a pen or coordinating different body parts, the cerebellum in the brain performs essential functions for controlling our movement. Researchers at the Institute of Science and Technology Austria (ISTA) investigated how a crucial set of synapses between neurons within it functions and develops. Their findings have now been published in the journal Neuron.
Even if you do not think about it, every day you are using the intricate circuits of neurons in your brain to perform astonishingly delicate movements with your body. One essential unit in this is the cerebellum playing a key role in fine motor control, coordination, and timing.
“Every time I go climbing, I make use of complex structures in the connections between neurons—the synapses—in my cerebellum. In this project, we wanted to understand how they actually work,” Peter Jonas, Magdalena Walz Professor for Life Sciences at the Institute of Science and Technology Austria (ISTA), relates his research to his everyday experiences. In a new study, now published in the journal Neuron and led by PhD student Jingjing Chen in Jonas’ research group and Walter Kaufmann from ISTA’s Electron Microscopy Facility with crucial support from ISTA’s Professor Ryuichi Shigemoto, the scientists unraveled some of the key mechanisms in the synapses of the cerebellum.
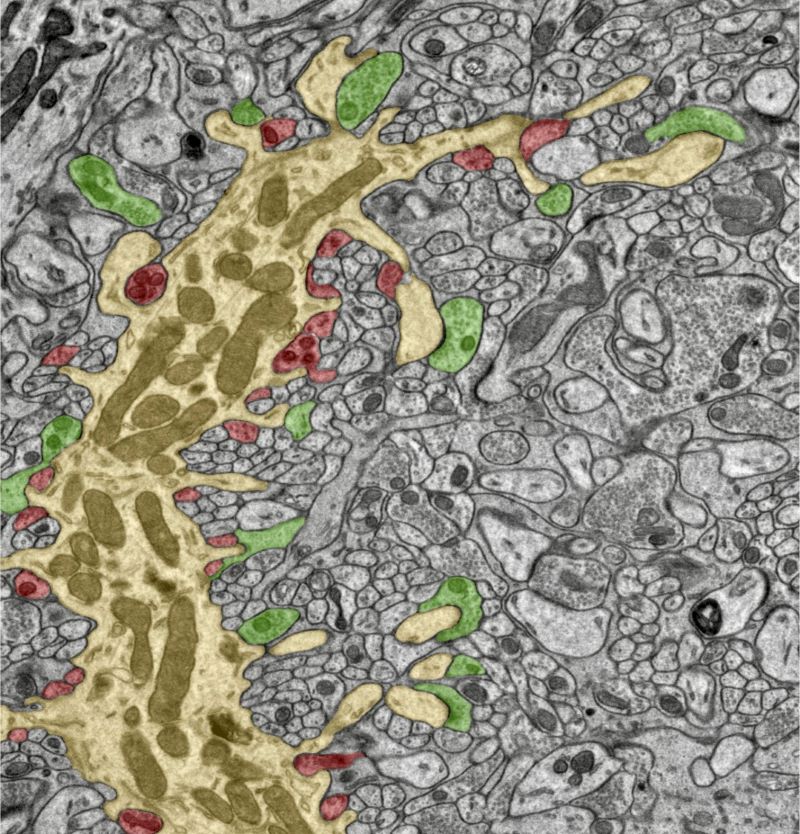
Controlling synapses
“The cerebellum receives a lot of inputs from the other parts of the brain and from sensory systems but its output signals controlling movement all go through a critical kind of neuron called Purkinje cell,” Chen explains. “And in turn all these Purkinje cells each get a lot of signals from other neurons in the cerebellum through their input synapses. We looked at a specific kind of synapse that inhibits the activity of the Purkinje cells and plays a crucial role in controlling their output signals.”
The Purkinje cells in the cerebellum form a bottleneck for its motor control signals, but many aspects of how this is achieved on a molecular and cellular level have remained unclear. In their study, the researchers have now uncovered the details of the cells’ inhibitory synapses.
The researchers used subcellular patch clamp recording, guided by an advanced light microscopy technique called online confocal imaging, to look at the function of these synapses in great detail. In parallel, they used electron microscopy to study the structure of the synapses at highest possible resolution. Some of the features they were looking at are just a few nanometers—millionths of a millimeter—in size. Chen and her colleagues measured a variety of parameters of the synapses, like where and how often neurotransmitters are being released, as well as the size of the tiny bubbles that contain the neurotransmitters in order to build a computer model of the whole process. They did this with neurons from brains of mice at different ages to understand their development over time.
Jonas, who developed the computer model, adds, “Building a model lets you properly understand a system and also sparks new ideas for future experiments. However, you have to be careful and keep it grounded in reality with experimental data.”
With their computer model simulating what is going on at different stages of development from early to adult age, the scientists could see how the inhibitory synapses of the Purkinje cells in the cerebellum delicately influence the cell’s signal output allowing for fine control of motor skills.
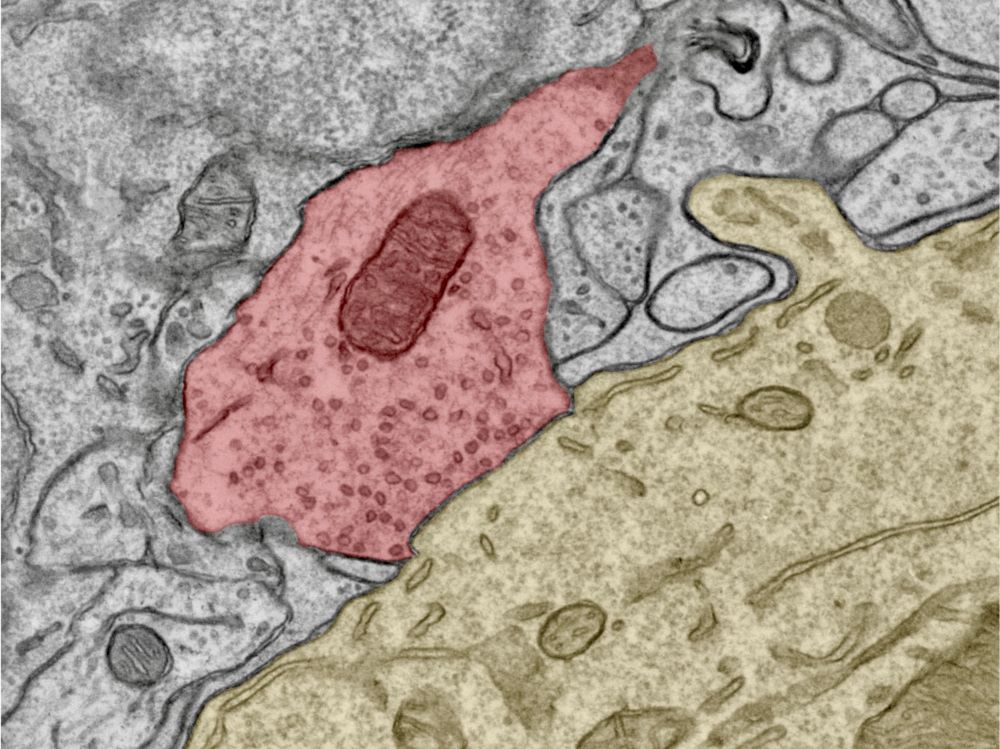
Emerging structure
“We found that, at an early age, all the mechanisms in the synapse are quite randomly organized and are not that exact in their functions,” Kaufmann, who joined ISTA as one of the first staff scientists about ten years ago, explains the results. “As the circuit matures, the synapse becomes more structured, approaching a point-to-point configuration, and thereby reaches a higher level of functional precision”. Chen, who wants to continue her work on the cerebellum after her PhD, adds, “The tricky thing was that we cannot just ask the cells directly how they work. Instead, we have to take snapshots—meaning measurements—at different stages of development and deduce the processes and overall structure behind them.”
Understanding this kind of synapse is not just a worthwhile endeavor in fundamental science, but, in the future, may even help researchers to address neurological diseases whose cause is indicated to lie in a malfunctioning of synapses in the brain.
Jonas adds, “This project created a bridge between the biophysical properties of synapses and what we can actually observe under the microscope. It is a great step forward in understanding the details of how the cerebellum helps us control and coordinate our movement. Initially, the required experiments seemed impossible, but with the support of Ryuichi Shigemoto, as well as our dedicated colleagues from the Jonas group and the scientific service units, we managed to bring our understanding of these crucial synapses to the point.”


Publication:
Chen J., Kaufmann W., Chen C., Arai I., Kim O., Shigemoto R., and Jonas P. 2024. Developmental transformation of Ca2+ channel–vesicle nanotopography at a central GABAergic synapse. Neuron. DOI: 10.1016/j.neuron.2023.12.002
Funding information:
This research was supported by the Scientific Services Units (SSUs) of ISTA (Electron Microscopy Facility, Preclinical Facility, and Machine Shop). The project received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (grant agreement No 692692), the FWF – Fonds zur Förderung der Wissenschaftlichen Forschung (Z 312-B27, Wittgenstein award; P 36232-B), all to Peter Jonas, and a DOC fellowship of the Austrian Academy of Sciences to Jingjing Chen.
Information on animal studies:
To understand how the structure and function of synapses in the brain changes during development, it is essential to study them in intact brain tissue. No other methods, such as cell culture or in silico models, can serve as alternatives. The animals were raised, kept, treated, and euthanized according to the strict regulations of Austrian law. Experimental procedures were discussed and approved by the institutional ethics and animal welfare committees at ISTA in accordance with best scientific practice guidelines and national legislation.



