November 7, 2014
Watching the neocortex develop
IST Austria Professor presents new findings relevant for our understanding of brain development in the current issue of Cell • Hippenmeyer and co-authors apply Mosaic Analysis with Double Markers (MADM) to decode how neocortical projection neurons emerge • MADM method permits quantitative look at cell division patterns of single stem cell progenitors at unprecedented resolution
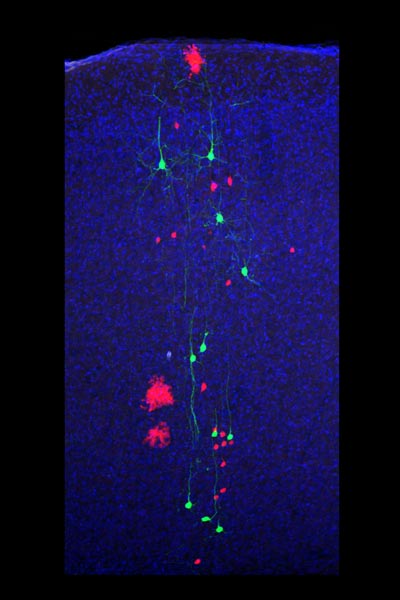
The neocortex is the biggest part of the human brain, and controls all higher cognitive functions such as conscious thought, control of movement, and sensory perception. In a paper published in Cell on November 6, Simon Hippenmeyer, Assistant Professor at IST Austria, presents how neurons and glial cells are formed in the developing neocortex at an unprecedented resolution. For this study, Simon Hippenmeyer’s research group worked closely together with Ekaterina Papoucheva from the Bioimaging Facility at IST Austria, the lab of Songhai Shi at the Sloan Kettering Institute (NY, USA), and Liqun Luo at Stanford University (CA, USA), as well as with physicist Benjamin Simons at Cambridge University (UK) and computer scientist Kun Huang at Ohio State University.
The neocortex builds up from the inside out. Neurons are produced when radial glial progenitors (RGPs) divide. Early-born neurons stay close to the layer of RGPs, while neurons born later migrate past early-born neurons to form more superficial layers. Early- and late-born neurons also differ in their function: While early-born neurons mostly connect to other brain areas outside the neocortex, later-born neurons preferentially connect with other neurons within the neocortex in the same or the opposite hemisphere. So far, it has been unclear how RGPs divide to produce all the different cortical neuron types. Simon Hippenmeyer and co-authors used the Mosaic Analysis with Double Markers (MADM) technique to decipher the principles of excitatory neuron genesis in the neocortex of mice at single cell resolution. The MADM technique provides an unprecedented approach that allows the researchers to quantitatively look at the cell division patterns of stem cell progenitors and trace the precise lineage of individual RGPs. The authors show that RGPs follow a remarkably deterministic and coordinated developmental program. All RGPs divide symmetrically at first, which produces a greater pool of progenitor cells. After a while, symmetric proliferation decreases and RGPs switch to an asymmetric division mode that generates neurons. Intriguingly, upon entry into the neurogenic phase, individual RGPs produce a defined unit of about 8-9 neurons indicating a unitary output. Preceding symmetric RGP divisions seem to amplify this unit and excitatory neurons in the neocortex thus appear to be produced in ‘quanta’.
All RGPs produce neuronal units that include distinct types of neurons – early-born, deep layer neurons and later-born, superficial layer neurons. Simon Hippenmeyer and colleagues so provide clear evidence for a model whereby individual RGPs are multipotent and thus can contribute to the pools of most if not all major types of cortical projection neurons.
The researchers also studied what happens when they remove the transcription factor OTX1, which binds to DNA and regulates which genes are turned on or off. OTX1 is expressed and active in RGPs at the early stage of neuron production. Therefore, it had been assumed that OTX1 plays a role only in the production of early-born neurons. Simon Hippenmeyer and colleagues now show that removing OTX1 leads to a reduction of both early- and late-born neurons: More generally, these findings might also have implications for our understanding of neurodevelopmental diseases since mutations in OTX1 cause severe microcephaly and epilepsy and have been associated with autism.
Lastly, the authors demonstrate that upon the completion of neurogenesis, individual RGPs proceed to gliogenesis at a predictable rate. The deterministic mode of both neuron and glia production may play a crucial role in dictating the correct ratio of neurons to glia and could therefore be critical for overall neocortex assembly and function. In future studies, it will be interesting to quantitatively assess the stem cell behavior and unit size of RGPs in different species and determine their relationship to neocortical expansion in the course of evolution.



